Growth and Characterization of L-Proline Doped Adp Crystals for Optoelectronic Applications
M. Iyanar1*, J. Thomas Joseph Prakash2, C. Muthamizhchelvan3, S. Ekadevasena4, J. Martin Sam Gnanaraj4, S. Shek Dhavud5
1PG and Research Department of Physics, National College, Trichy
2Department of Physics, Government Arts College, Trichy
3Department of Physics, SRM University, Kancheepuram 603 203
4PG and Research Department of Physics, H. H. The Rajah’s College, Pudukkottai
5PG and Research Department of Physics, Jamal Mohammed College, Trichy
DOI : http://dx.doi.org/10.13005/msri/120104
Article Publishing History
Article Received on : 31 Dec 2014
Article Accepted on : 27 Jan 2015
Article Published : 27 Jan 2015
Plagiarism Check: Yes
Article Metrics
ABSTRACT:
The present work is a comparative analysis of the properties of undoped and L-Proline doped ADP crystals. Slow evaporation solution growth technique was employed for growing undoped and L-Proline doped ADP crystals. The unit cell parameters were estimated by single crystal X-Ray diffraction analysis. The crystalline nature of the samples was revealed by Powder X-Ray diffraction analysis. The presence of functional groups and the spectral properties was identified by FTIR spectral analysis. The optical property of the material was examined by UV-Visible spectral analysis. The surface morphology and the elemental composition of the material was studied by SEM and EDS analysis. The nonlinear optical property of the samples was tested by Kurtz and Perry experimental setup.
KEYWORDS:
Solution Growth; FTIR; UV; SEM; EDS
Copy the following to cite this article:
Iyanar M, Prakash J. T. J, Muthamizhchelvan C, Ekadevasena S, Gnanaraj J. M. S, Dhavud S. S. Growth and Characterization of L-Proline Doped Adp Crystals for Optoelectronic Applications. Mat.Sci.Res.India;12(1)
|
Copy the following to cite this URL:
Iyanar M, Prakash J. T. J, Muthamizhchelvan C, Ekadevasena S, Gnanaraj J. M. S, Dhavud S. S. Growth and Characterization of L-Proline Doped Adp Crystals for Optoelectronic Applications. Mat.Sci.Res.India;12(1). Available from: http://www.materialsciencejournal.org/?p=1483
|
Introduction
The materials with high optical nonlinearities are the target to the researchers since their applications in optoelectronics, telecommunication industries, laser technology and optical storage devices are innumerable. Ammonium dihydrogen orthophosphate (ADP) and potassium dihydrogen orthophosphate (KDP) are two of the oldest crystals grown in large size for many applications and continue to be interesting materials both academically and industrially [1-6]. ADP crystals have wide range of applications in integrated and nonlinear optics because of their piezo-electric (antiferroelectric at low temperature) and nonlinear optical property. Due to its interesting electrical and optical properties, structural phase transitions, and ease of crystallization, it has been the subject of a wide variety of investigations for several years [7, 8].
Ammonium dihydrogen Phosphate (ADP) is a representative of hydrogen bonded materials that possesses excellent dielectric, piezoelectric, antiferroelectric, electro-optic and nonlinear optical properties. Growth and studies of ammonium dihydrogen phosphate is a center of attention to researchers because of its unique properties and wide applications. Single crystals of ADP are used for frequency doubling and frequency tripling of laser systems, optical switches in inertial confinement fusion and acousto-optical devices. ADP crystallizes in a body centered tetragonal structure with the space group of I 4 2d and its unit cell parameters are a = b = 7.510 Å and c = 7.564 Å [9, 10].
Amino acids are interesting materials for NLO applications as they contain a proton donor carboxyl acid (-COOH) group and proton acceptor amino (-NH2) group in them [11].
Amino acid family crystals are playing an important role in the field of non-linear optical organic molecular crystal [12]. The effect of additives as amino acid on growth, habit modification and different structures of ADP crystals has been studied [13, 14]. It was also re-ported that the addition of some of the amino acids as dopant enhances the nonlinear optical and piezoelectric properties of inorganic materials [14, 15].
Several researchers have carried out a lot of studies in pure and doped ADP [16-19]. DL-malic acid was successfully doped with ADP and found that the dopant can affect the various properties of ADP [17]. Ammonium chloride doped ADP crystals was found mechanically harder and thermally more stable than pure ADP [18]. NLO single crystals with high conversion efficiencies for second harmonic generation are desirable in various applications. With the aim of discovering new useful materials for academic and industrial use, an attempt has been made to modify ADP crystals by adding some amino acids [20], [21] and [22]. Since most of the amino acids exhibit NLO property, it is expected that the addition of some amino acids, such as l-arginine, in ADP could also improve the various properties [19]. On the other hand, uniaxial solution crystallization method of Sankaranaranyanan-Ramasamy is a novel method to grow single crystals with 100% solute-crystal conversion efficiency [23], [24] and [25]. Recently many papers show that the SR method grown crystals have higher crystalline perfection than conventional grown crystals [19], [20] and [21]. L Proline doped ADP crystals were grown by T. Josephine Rani et. al., [26]. The work discusses the effect of L-Proline in altering the linear and nonlinear optical properties. The present work comprises of the influence of amino acid on the structural, optical, thermal and microstructural morphology of Ammonium dihydrogen phosphate crystals. The grown crystals were characterized by single crystal and powder XRD, FTIR, UV–vis NIR, SEM, EDAX Vicker’s microhardness and NLO studies to reveal their structures, spectral properties, optical transmittance, microstructural morphology, elemental composition, mechanical strength and SHG efficiency.
Synthesis and Crystal Growth
The starting materials used in the present work were of high purity (Merck, 99%) and of analytical reagent grade. Commercially available Ammonium Dihydrogen Phosphate (NH4H2PO4) and L-Proline (C5H9NO2) were used as precursors. Saturated solution of undoped ADP was prepared by conventional slow evaporation solution growth technique. 0.1 mol % L-Proline was added to the saturated ADP solution and the mixture was stirred for 5 hours using a magnetic stirrer at room temperature. A clear, transparent solution was obtained after 5 hours of continuous stirring. The solution was filtered in a clean beaker and kept covered with a perforated foil sheet. Transparent colorless crystals were obtained after a time span of 30 days. The photograph of as grown L-Proline doped ADP crystal is shown in Fig. 1
Results and Discussion
Single Crystal X-Ray Diffraction
The grown crystals were subjected to single crystal X-Ray diffraction for analyzing the unit cell parameters. ENRAF NONIOUS CAD4 X-Ray diffractometer was used for estimating the cell parameters of both undoped and L-Proline doped ADP crystals. L-Proline doped ADP crystals were found to crystallize in tetragonal symmetry with the space group of I42d. The unit cell parameters of L-Proline doped ADP crystal is a=b=7.521 Å, c=7.557 Å, α=β=γ= 90 ° Volume of the unit cell, V= 427.46 Å3. The obtained values coincide very well with the reported literature thereby confirming the structure. The slight variation in the unit cell values confirms the incorporation of additive into the crystal lattice of ADP crystals.
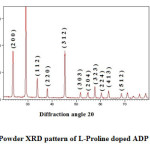
Powder X-Ray Diffraction
Well powdered L-Proline doped ADP crystals were subjected to Powder X-Ray diffraction analysis for analyzing the crystalline nature of samples. Powder X-Ray diffraction was performed using JEOL JDX 6030 Powder X-Ray diffractometer equipped with CuKα radiation (λ= 1.54056 Å). The indexed X-ray powder diffraction pattern of 0.1 mol % L-Proline added ADP crystals is shown in Fig. 2. The sharp peaks indicate the crystallinity of the grown crystals. The observed prominent peaks are (2 0 0), (1 1 2), (2 2 0), (3 0 3) and (3 1 2). The above mentioned changes are due to the presence of the additive L-Proline into the lattice of ADP crystal. However, there are no other phases emerging besides the tetragonal system which denotes that the incorporation of dopant does not change the structure of ADP crystal. The observed results are in good agreement with the reported values. The unit cell parameters of L-Proline added ADP crystals were calculated using JCPDS data as a=b=7.524 Å, c= 7.560 Å, Volume=427.97 (Å) 3.
Spectral Analysis of L-Proline Doped ADP Crystals
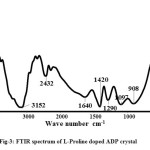
The spectral properties and the presence of the functional groups of L-Proline doped ADP crystals were identified by subjecting the samples to Fourier Transform Infrared analysis. FTIR analysis was carried out using PERKIN ELMER FTIR RXI Spectrophotometer and the spectrum was traced within the wavenumber range of 400 cm-1 to 4000 cm-1. The traced FTIR spectrum of L-Proline doped ADP crystals is shown in Fig. 3. The spectral vibrations and the corresponding assignments are tabulated in Table 1.
The effect of L-Proline on the vibrational frequencies of the functional groups of pure ADP crystal has been identified in the spectra. In the spectrum of ADP, the broad band around 3150 cm-1 is due to the O-H vibrations of water, P-O-H group and N-H vibrations of ammonium. The band at 2431 cm-1 is assigned to hydrogen bond [8]. The broadness is due to the hydrogen bonding interaction with adjacent molecules. The bending vibrations of water give the peak at 1633 cm-1. The peak at 1418 cm-1 is due to the bending vibrations of ammonium. The peak at 1285 cm-1 is due to the combination of the asymmetric stretching vibration of PO4 with lattice. The peaks at 1093 cm-1 and 907 cm-1 represent P-O-H stretching vibrations. The peaks at 562 cm-1 and 450 cm-1 are due to the PO4 vibrations.
In the spectrum of L-Proline added ADP crystal, the broad band appearing at 3152 cm-1 includes O-H vibrations of water and N-H vibrations of ammonium and L-Proline. The sharp peak at 1290 cm-1is due to CH2 wagging vibration of L-Proline.
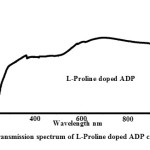
Linear Optical Analysis
The linear optical properties of L-Proline doped ADP crystals are examined by subjecting the materials to UV-Visible spectral analysis. The spectrum was traced between the wavelength ranges of 190 nm to 1100 nm. It was observed that L-Proline doped ADP crystals are transparent in the entire visible region. The observed transmittance value from the UV spectrum (Fig. 4) for L-Proline doped ADP crystal is 82%. Since the lower cut off wavelength falls within 300 nm L-Proline doped ADP crystals are efficient candidates in the fabrication of optoelectronic devices. The slight absorption peaks denote the presence of microstructural defects in the grown crystals.
Microstructural Morphology
The microstructural morphology of the undoped and L-Proline doped ADP crystals were obtained by subjecting it to Scanning Electron microscopy (SEM) analysis. JEOL 6360 TESCAN Scanning electron microscope was used for this process. The samples were scanned at various magnifications and different SEM micrographs were taken. The SEM micrograph of L-Proline doped ADP crystals is shown in Fig. 5. From the SEM micrographs of L-Proline doped ADP crystal the surface appears like a disordered heap. L-Proline is sticked to the surface of ADP crystals which is clearly seen in the SEM micrograph.
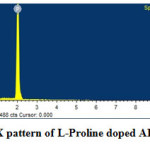
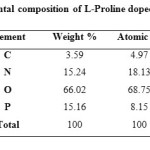
Elemental Composition
The elements present in the sample are analyzed by subjecting the samples to Energy Dispersive Spectral analysis. The measurements were carried out at room temperature with the help of JEOL 6360 Scanning Electron Microscope. Figure 6 shows the obtained EDX pattern. Elements present in the doped sample was identified and presented in terms of Weight% in Table-2. The variation in the weight percent of carbon is due to the addition of L-Proline into the crystal lattice f ADP crystal.
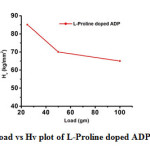
Vicker’s Microhardness Test
The microhardness characterization is extremely important as far as the device fabrication is concerned. The grown crystals were used for microhardness measurement using a Vickers microhardness tester fitted with a diamond indenter. The Vickers micro hardness number Hv of the crystal was calculated using the relation
Hv= 1.8544 P/d2 kg/mm2
Where P is the applied load in kg and d is the length of indentation impression in millimeter and 1.8544 is a constant of a geometrical factor for the diamond pyramid and the load Vs Hv graph is shown in Fig.7. The Vicker’s hardness number decreases with the increase in load.
Nonlinear Optical Studies
Kurtz and Perry powder technique is extremely useful for the initial testing of materials for second harmonic generation (SHG). The fundamental beam of wavelength 1064 nm, from a Q-switched Nd: YAG laser was used to test the second harmonic generation property of the grown crystals. The pure and L-Proline added ADP crystals were ground into fine powder and packed in micro capillary tubes mounted in the path of laser pulses with pulse width 6 ns and repetition rate 10 Hz, having an input energy of 0.68 mJ/pulse. The second harmonic generation was confirmed by the green emission of wavelength 532 nm from the samples. The output energy for 0.1 mol% L-Proline added ADP samples was measured to be 5.7 mJ/pulse. This SHG efficiency of ADP with the addition of L-Proline is due to the fact that L-Proline has NH3+ and COO − groups. The optically active amino group may get added in the ADP structure and increase its non-centrosymmetry thereby increasing its SHG efficiency.
Conclusion
L-Proline doped ADP crystals were grown by conventional slow evaporation solution growth technique. The structure was confirmed by single crystal x-ray diffraction. The crystallinity of the undoped and doped crystals was confirmed from the powder x-ray diffraction pattern. The presence of functional groups and the spectral properties were assessed by FTIR spectrum. The optical properties such as the lower cut off and % of transmittance was analyzed from the UV visible spectral analysis. The surface morphology of the undoped and doped materials were studied using SEM analysis. The incorporation of amino acid was confirmed by EDS analysis. The NLO studies confirm the SHG efficiency of the L-Proline added ADP crystal. The results suggest that L-Proline added ADP crystals are potential candidates in NLO device applications.
References
- Yokotani, T. Sasaki and K. Yamanaka. Appl. Phys. Lett., 48, pp. 1030, (1986).
CrossRef
- H. Tukubo and H. Makita. J. Cryst. Growth, 94, 469, (1989).
CrossRef
- D. Xu and D. Xue. J. Cryst. Growth, 310, 1385, (2008).
CrossRef
- X. Ren, D. Xu and D. Xue. J. Cryst. Growth, 310, 2005, (2008).
CrossRef
- D. Xu and D. Xue. J. Cryst. Growth, 310, 2157, (2008).
CrossRef
- D. Xu and D. Xue. J. Cryst. Growth, 286, 108 (2006).
CrossRef
- J.D. Lindl. Phys. Plasmas, 2, 3933, (1995).
CrossRef
- D. Eilmerl. Ferroelectrics, 72, 95 (1987).
CrossRef
- N. Zaitseva, L. Carman, Prog. Crystal Growth Charact. 43 1(2001)
CrossRef
- L. Tenzer, B.C. Frazer, R. Pepinsky, Acta Cryst. 11 505 (1958)
CrossRef
- P.Selvarajan, J.Glorium Arul Raj, S.Perumal, J. Crystal Growth 311, 3835,(2009).
CrossRef
- S. R. Marder, B. G. Tiemann, J. W. Perry, Ameri-can Chemical Society, Washington, 280 (1991).
- P. V. Dhanaraj, G. Bhagavannarayana and N. P. Rajesh, Materials Chemistry and Physics, 112, 490, (2008).
CrossRef
- M. Meena and C. K. Mahadevan, Cryst. Res. Tech, 43, 166 (2008).
CrossRef
- K. D. Parikh, D. J. Dave, B. B. Parekh, and M. J. Joshi, Cryst. Res. Tech, 45, 1, (2010).
CrossRef
- P. Rajesh and P. Ramasamy. Spectrochim. Acta A, 74, 210 (2009).
CrossRef
- P. Rajesh and P. Ramasamy. J. Cryst. Growth, 311, 3491 (2009).
CrossRef
- P. Rajesh and P. Ramasamy. Mater. Lett., 63, 2260 (2009).
CrossRef
- M. Meena and C.K. Mahadevan. Cryst. Res. Technol., 43 166 (2008).
CrossRef
- F. Zhang and D. Xue. Modern Phys. Lett. B, 23, 3943 (2009).
CrossRef
- D. Xue and H. Ratajczak, J. Mol. Struct., 716, 207 (2005).
CrossRef
- Z. Latajka, G. Gajewski, A.J. Barnes, D. Xue and H. Ratajczak, J. Mol. Struct., 928 121 (2009).
CrossRef
- K. Sankaranarayanan and P. Ramasamy. J. Cryst. Growth, 280, 467 (2005).
CrossRef
- K. Sankaranarayanan. J. Cryst. Growth, 284, 203(2005).
CrossRef
- K. Sankaranarayanan and P. Ramasamy. Cryst. Res. Technol., 41, 225 (2006).
CrossRef
- T. Josephine Rani, Fernando Loretta, P. Selvarajan, S. Ramalingom, S. Perumal Recent Res. Sci .Tech, 3, 69 (2011).
Views: 2,481
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Material Science Research India An International Peer Reviewed Research Journal
Material Science Research India An International Peer Reviewed Research Journal