Anima Upadhyay and M. Chandrakala
Department of Chemistry, Sir MVIT, Bangalore, Karnataka, India. 562157
Corresponding Author Email: Animaupadhyay@gmail.com
DOI : http://dx.doi.org/10.13005/msri/130207
Article Publishing History
Article Received on : 24 Sep 2016
Article Accepted on : 17 Nov 2016
Article Published : 26 Nov 2016
Article Metrics
ABSTRACT:
The corrosion on smooth tinned iron material has been studied in different media. Solutions of sulphuric acid, hydrochloric acid, acetic acid and salt solutions of ammonium chloride and sodium chloride were prepared to investigate the corrosion on the metal plates. The plates was immersed in the solutions for a period of time same for all the solutions in order to determine the most aggressive environment. Weight loss technique was adopted to study the rate of corrosion. The result reveals that the material got corroded in this order sulphuric acid > hydrochloric acid > acetic acid > ammonium chloride > sodium chloride. It was therefore concluded that the rate of corrosion was high in strong acids compared to the weak acids and salts.
KEYWORDS:
Acids; Corrosion rate; salts; Tinned iron; Weight loss method
Copy the following to cite this article:
Upadhyay A, Chandrakala M. Corrosion Studies on Tinned Iron Plates from Infant Food Cans. Mat.Sci.Res.India;13(2)
|
Introduction
Corrosion is a natural phenomenon. Destruction of the metal due to its slow oxidation with the surrounding environment is called corrosion. The wet chemical theory explains the corrosion of metals / alloys in various mediums / environment. Corrosion and its rate depend upon many factors. Surface and nature of the material play a significant role in corrosion and is a determining factor in the rate of corrosion. Presence of air, moisture, salts, free ions, gases (mainly acidic gases) affects the rate of corrosion and may lead to excessive corrosion. Recently there is a revolution in the medical field where artificial organs are implanted in the body if the original organs fail to function. These artificial organs are generally made up of metals and alloys. These artificial organs should possess high corrosion resistance otherwise the patients would suffer severe effects caused by the corrosion of the implanted organs. Modernization started the growth of Industries and cities. In this growth, infrastructure has played a major role. All this require various kinds of alloys, metals and composite materials which due to corrosion requires heavy monetary maintenance. Hence it is a matter of concern therefore many authors have studied the role of environment on the corrosion of the metal by weight loss method.1,22
The other drawback of the modern age is that the people are busy in their hectic schedule and finding it difficult to cook food every day, therefore they depend upon the readymade food stuff. This generally comes in packed cans made of tinned iron. Thus there is an extensive use of metals and alloys in our day to day life compared to the people in the past. Hence the study of corrosion has become the need of the day. If the rate of corrosion is studied prior to its usage we can save ourselves from large damages that could ruin our lives due to corrosion effects.
Corrosion can be explained with the help of electrochemical theory which explains corrosion as the formation of galvanic cells. These cells have anodic and cathodic sites. Due to the interaction of the metal with the environment, the metal at the anode starts corroding resulting into the loss of the metal.
Materials and Methods
Sample Preparation
Square tinned iron (3cm× 3cm) metal plates were used as sample. Nestle (Lactogen infant powder tinned iron cans were used preparation of the sample. A small hole near the upper edge of the plate was made for hanging in test media. Each plate was cleaned, degreased and washed by double distilled water followed by acetone and dried.13
Test solution
2% solutions of H2SO4, HCl, CH3COOH, NH4Cl, NaCl were prepared using double distilled water. All reagents used were of analytical grade (E – Merck Millipore’s EMSURE ®).
Weight loss measurements
The weight loss experiments were carried out using surface treated square specimens of tinned iron having surface area 9 sq.cm. These were immersed in beakers containing 50ml of above prepared corrosive solutions for 24 hours at room temperature. Weight (W1) of the plates before immersion was recorded. After 24 hours of immersion the plates were removed and washed with double distilled water followed by acetone. They were then dried and weighed (W2). Difference in the weight, gave the loss in weight of the material (W). (Table 1)
Weight Loss Method
It is a simple, accurate and reliable method for the study of corrosion of metals / alloys as studied by many authors.4,7,9 In this method the metal of known area is exposed to the environment for a definite period of time and the difference in the weight before and after the exposure is calculated. The rate of corrosion is calculated as follows:
Weight loss (W) X Constant (K)
Corrosion rate (mmpy) = ───────────────────────────────────────
Exposed area (A) X Density of the specimen (D) X Exposure time (T)
Where, K = 8.76 X 10 4 (constant), W = weight loss in g, A = area in sq.cm and D = 7.78 gm / cm3 (density), T = exposure time in hours.
Result and Discussion
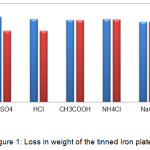
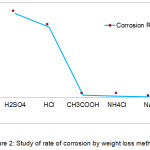
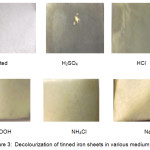
The results obtained from the study suggest that the maximum weight loss was in the sulphuric acid followed by hydrochloric acid and acetic acid. In ammonium chloride the loss in weight was very little and almost none in sodium chloride. (Table 1, Fig 1). On the basis of weight loss the corrosion rate was calculated which was then used to predict the severity of corrosion on the metal [2]. The rate of corrosion was in the following order sulphuric acid > hydrochloric acid > acetic acid > ammonium chloride > sodium chloride. (Table 1, Fig 2). The rates of corrosion infer the severity of corrosion on the material. In sulphuric and hydrochloric acid the corrosion rate was found to be severe, in acetic acid and ammonium chloride it was moderate where as in sodium chloride it was found to be safe (Table 1). The pictures of affected plates of tinned iron in the above media also greatly depict the corrosion by showing decolourization and tarnishing effect (Fig 3). Maximum decolourization was found in sulphuric and hydrochloric acid. It has almost turned black. In acetic acid and ammonium chloride effect of tarnishing is less than the strong acids. Sodium chloride showed very little tarnishing.
The probable chemical reaction that would have caused in the various acid are given below
- Fe + HCl → FeCl2 + H2↓
- Fe + H2SO4 → FeSO4 + H2↓
- Fe + CH3COOH → Fe(CH3COO)2 + H2↓
In all the above cases, liberation of hydrogen leads to corrosion.
Ammonium chloride is a weak electrolyte hence, the dissociation of electrolyte is almost negligible. Therefore the corrosion is very little.
Table 1: Corrosion rate by weight loss method
|
Solutions
employed
|
Initial sample weight (W1)
|
Final sample weight (W2)
|
Weight loss
W = (W1 – W2)
|
% of weight loss =
(W / W1) X 100
|
Corrosion Rate (mmpy)
|
Quality according to rate
|
|
H2SO4
|
1.5346
|
1.2820
|
0.2526
|
16.46
|
13.1675
|
Severe
|
|
HCl
|
1.4901
|
1.2782
|
0.2119
|
14.22
|
11.0459
|
severe
|
|
CH3COOH
|
1.4752
|
1.4690
|
0.0062
|
0.420
|
0.3231
|
moderate
|
|
NH4Cl
|
1.4886
|
1.4857
|
0.0029
|
0.194
|
0.1511
|
moderate
|
|
NaCl
|
1.4428
|
1.4423
|
0.0005
|
0.034
|
0.0260
|
safe
|
Figure 1: Loss in weight of the tinned Iron plates
Figure 2: Study of rate of corrosion by weight loss method
Figure 3: Decolourization of tinned iron sheets in various medium
Conclusion
The authors suggest that keeping the processed and cooked food stuff in the tinned iron cans is not a good option especially the baby food. If these materials are employed for packaging the food, they should be kept with utter care and away from the corrosive environment. Precaution should be taken and the food should not be consumed a week before the expiry date. Doing so, the food will be prevented from getting stale and poisonous. Further studies on this topic are needed on inhibitors with practical implication of slowing down the corrosion process, particularly in the food packaging.
Acknowledgement
Authors extend thanks to the institution for providing infrastructure to conduct the study.
References
- Aminu D. Usman and Linus N. Okoro., Chem Sci Rev Lett. 2015;4(13):17–24.
- S. SYED., Emirates Journal for Engineering Research. 2006;11(1):1-24.
- Kingsley O. Oparaodu and Gideon C. Okpokwasili., International Journal of Environmental Bioremediation & Biodegradation. 2014;2(5):243-249.
- V. R. Rathi, S. D. Nirmal and S. J. Kokate., J. Chem. Pharm. Res. 2010;2(2):97-100.
- Anyanwu Samuel Ikechukwu, Eseonu Obioma, and Nwosu Harold Ugochukwu., The International Journal Of Engineering And Science. 2014;3(10):48-60.
- O. O. Ajide and K. W. Agara., International Journal of Metallurgical Engineering. 2012;1(1):7-11.
CrossRef
- Mamatha.G.P., Pruthviraj. R .D and Ashok. S. D., International Journal of Research in Chemistry and Environment. 2011;1:85-88.
- B. Bobić, S. Mitrovic, M. Babic and I. Bobić., Tribology in industry. 2009;31(3&4).
- H. Zarrok 1, H. Oudda 1, A. Zarrouk 2, R. Salghi 3, B. Hammouti 2, and M. Bouachrine., Der Pharma Chemica. 2011;3(6):576-590.
- Chinwko Emmanuel Chuka, Odio B. O., Chukwuneke J. L., and Sinebe J. E., International Journal Of Scientific & Technology Research. 2014;3(7).
- Ehteram A Noor and Aisha Al-Moubaraki., International journal of electrochemical science. 2008;3(7):806-818.
- Osarolube, E, Owate, I. O. and Oforka, N. C., Scientific Research and Essay. 2008;3(6):224-228.
- Guddi Choudhary, Arpita Sharma and Alka Sharma., International Journal of Innovative Research in Science, Engineering and Technology. 2013;2(10).
- Ya Zhang, Sen Wu, Qiurong Chen, Xuehua Zhou and Zhongling Wei., Int. J. Electrochem. Sci. 2015;10:1015–1026.
- Ikpesu and Jasper Ejovwokoghene., Journal of Research in Environmental Science and Toxicology. 2014;3(1):15-20.
- Geetha Mable Pinto, Jagannath Nayak and A Nityananda Shetty., Int. J. Electrochem. Sci. 2009;4:1452–1468.
- R. Selva Kumar and V. Chandrasekaran., Material Science Research India. 2015;12(1):68-78.
CrossRef
- A. Sahaya Raja, P. Angel, R. Sonisheeba, J. Thomas Paul raj, S. Sivakumar, R.Venkatesan and J. Sathiyabama. International Journal of Advanced Research in Chemical Science. 2014;1(1):10-21.
- J. Hemalatha, A. Sankar, S. Ananth Kumar and S. Ramesh Kumar., International Journal of Computer Engineering & Science. 2013;3(1):15-20.
- S. Khalid Hasan and Pinky Sisodia., Rasayan J. Chem. 2011;4(3):548-553.
- Radha Mishra., International Journal of Chemistry. 2014;1(1):18–21.
- A. Rajendran, C. Karthikeyan., International Journal of Plant Research. 2012;2(1):9-14.
CrossRef
Views: 655
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Material Science Research India An International Peer Reviewed Research Journal
Material Science Research India An International Peer Reviewed Research Journal