K. Pavani*1  , V. Ajay Kumar2
, V. Ajay Kumar2  and N. Laxman2
and N. Laxman2
1Department of chemistry, RGUKT-Basar, Telangana, India, 504107.
2Department of MME, RGUKT-Basar, Telangana, India, 504107.
Corresponding author Email: kpavani@rgukt.ac.in
DOI : http://dx.doi.org/10.13005/msri/140204
Article Publishing History
Article Received on : 26 Oct 2017
Article Accepted on : 23 Nov 2017
Article Published :
Plagiarism Check: Yes
Article Metrics
ABSTRACT:
This paper deals with the investigation of the reactions of aqueous molybdates in the presence of copper ions under varying pH to obtain new copper molybdates and rationalize the structures formed in terms of the self-assembling nano-building blocks of polyoxomolybdates (POMs). The reactions led to the precipitation of porous copper molybdates: 1D polymeric anions in (NH4)6[{Cu(H2O)4}{Mo36O112(H2O)16}].21H2O, 1, a 3D framework bimetallic oxide in Cu3Mo2O9, 2. Our results reported here clearly suggest that copper complexes formed during the initial stages of the reaction probably dictate the architecture of the final solids. Interestingly the magnetic properties studied on these compounds showed that 1 shows paramagnetic ordering since magnetically active centres i.e. Cu(II) are separated by large diamagnetic centres of {Mo36} cluster anions where as 2 shows the dominance of antiferromagnetic near-neighbor interaction.
KEYWORDS:
Crystal Structure; Giant Clusters; Polyoxomolybdates; Self-assembly;
Copy the following to cite this article:
Pavani K, Kumar V. A, Laxman N. pH as a Structure Director in the Synthesis of Copper Molybdates. Mat.Sci.Res.India;14(2)
|
Introduction
Polyoxomolybdate (POM) is an important class of inorganic materials that is currently attracting considerable attention due to its importance in the area of catalysis, sensors etc.1-4 The recent discovery of giant molybdenum clusters by Müller and his group5-8 has evoked remarkable interest to understand the self-assembly processes leading to the formation of POM clusters with varying size and shape with a variety of supramolecular assemblies thereby occurring in the solid state. A significant addition to this complex range of solids is the introduction of transition metal, which provides innumerable possibilities by coordinating linking the POM clusters into multidimensional structures. For construction of a specific architecture with one-, two- and three-dimensional networks such as molecular grids, bricks, ladders, rings, boxes, honeycombs, helicates etc.,9-11 design and selection of suitable metal ions are extremely crucial; it is also necessary to develop a rational synthetic protocol to engineer the structure of the final solid. Soft chemical routes have been adopted by several groups, in particular Zubieta et al12-16 to crystallize a large number of POMs from aqueous molybdate solution. Almost all groups have mainly employed hydrothermal/solvothermal reactions to prepare these solids. However, there is very little or no attempt to rationalize the synthetic procedure in terms of the reaction chemistry that is taking place in aqueous solution which dictates the fate of the final structure. The problem is also complicated by the black-box nature of the hydrothermal reaction and the dynamic nature of POM clusters that continuously change to smaller or larger fragments with varying pH, ionic strength and nature of metal ions.
Our group has been investigating the reaction of aqueous molybdates with a number of metal ions under varying pH in the presence of various organic amines that are linear, cyclic, aromatic and long chain surfactants with a view to obtain solids based on self-assembling nano-building blocks of POMs.17-20 In this work, we preferred cupric ions as a large number of structurally diverse copper molybdates are isolated from aqueous solution. Also, these can exhibit promising magnetic and catalytic properties. This paper is an attempt to examine the influence of pH on the products formed. Our results reported here clearly suggest that copper complexes formed during the initial stages of the reaction dictate the architecture of the final solids.
Experimental Section
Method
All the reagents were purchased from Aldrich and used as such without further purification. The samples prepared in this chapter are synthesized under slow evaporation. (NH4)6Mo7O24.4H2O (0.625mmol, 0.7724g) and CuCl2.2H2O (5mmol, 0.8524 g) were dissolved in 40 mL of distilled water and the pH was adjusted using dilute HCl. When pH was ~2, the compound 1 crystallized out after two weeks while the same solution at pH~6 yielded instantaneously an amorphous phase. This phase on heating at 300°C resulted in a single phasic solid.2
Characterization
Single crystal diffraction studies were carried out on a Bruker AXS SMART Apex CCD diffractometer with a MoKa (0.71073Å) sealed tube at 28°C. The software SADABSwas used for absorption correction and SHELXTL for space group and structure determination and refinements.21-22 The molybdenum atoms were located first and then remaining atoms were deduced from subsequent difference Fourier syntheses. The hydrogen atoms were located using geometrical constraints. All the atoms except H were refined anisotropically. The least-squares refinement cycles on F2 were performed until the model converged. Crystal data is provided in table 1.
The infrared spectra were recorded on a Nicolet 5DX spectrophotometer with pressed KBr pellets. TG and DT analyses were carried out using Perkin-Elmer TGA7 and DTA7 system on well ground samples in flowing nitrogen atmosphere with a heating rate of 10°C/min. Room-temperature X-ray powder diffraction data were collected on a Bruker D8 Advance diffractometer using Ni-filtered CuKa radiation. Data were collected with a step size of 0.02° and at count time of 2s per step over the range 2° < 2q < 60°. The susceptibility measurements as a function of temperature were performed with a SQUID magnetometer. Diamagnetic corrections were estimated using Pascal’s constants.
Table 1: Crystal data of (NH4)6[{Cu(H2O)4}{Mo36O112(H2O)16}].21H2O, 1
|
Crystal system
|
Monoclinic
|
|
Space group
|
C 2/c
|
|
T/K
|
300(2)
|
|
a /Å
|
39.848 (7)
|
|
b/Å
|
19.495 (4)
|
|
c/ Å
|
25.426 (5)
|
|
a/ o
|
90
|
|
b/ o
|
124.70(3)
|
|
g/ o
|
90
|
|
V/ Å3
|
16239.0(8)
|
|
Z
|
4
|
|
Collected reflections
|
53341
|
|
Unique reflections
|
8739
|
|
Observed reflections
|
7731
|
|
RI[I> 2s(I)]
|
0.0590
|
|
WR2(all)
|
0.1850
|
|
CCDC or ICSD Nos.
|
418439
|
Results and Discussion
(NH4)6[{Cu(H2O)4}{Mo36O112(H2O)16}].21H2O, 1
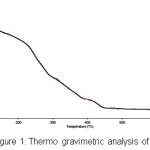
It is a single phase, as established by comparing experimental and simulated powder XRD patterns. FTIR spectrum of 1 exhibits four bands at 919, 879, 845, 645 cm-1 that are attributed to the vibration modes of n(Mo-Ot) and n(Mo-Ob) of the Mo36 cluster anion. Features at 3250, are characteristic absorptions of the ammonium ion. Peaks in the range 959–876 and at 850 cm-1 can be attributed due to the vibrations of Mo=O and Mo-O-Mo. In the TGA of 1 (Figure 1) the weight loss was observed in several steps where the first two steps were corresponding to loss of water molecules and the third step corresponds to the weight loss of ammonium ions. Latter weight loss corresponds to the degradation of the molybdates clusters.
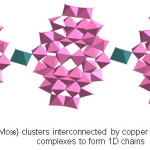
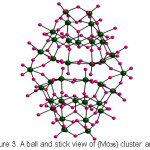
The crystal structure analysis of (NH4)6[{Cu(H2O)4}{Mo36O112(H2O)16}].21H2O, 1 showed the occurrence of 1D covalently bonded chains, built up from the polyoxomolybdate cluster containing thirty six molybdenum atoms linked through copper hydrate complex (Figure 2). Each Mo36 unit contains two Mo18 subunits built of sixteen edge-sharing and vertex-sharing MoO6 octahedra and two pentagonal bipyramids (Figure 3) that are in turn related by an inversion center. Among the 128 oxygen atoms of the cluster anion, 16 oxygens were water molecules as suggested by the bond valence sum calculations (Table 2). The Cu(II) sites exhibit octahedral geometry {CuO6} with four oxygen donors of water molecules on a basal plane and two bridging oxo groups of the Mo36 clusters at the apical position extending it into 1D infinite chain. The Cu-O distances [~2.513 (1) Ǻ] linking the anion clusters are much larger than Cu-O distances [~2.233 (3) and 2.179(1) Ǻ] on the basal plane. 1D chains made of Mo36 cluster cores have been reported earlier in few solids.23-25 The 1D chain observed in our case is similar to that of (NH4)6[Gd2Mo36O114(H2O)22].50H2O,26 where the Mo36 units are linked through Gd(H2O)6 polyhedra. However 1 is the first example wherein Mo36 units are linked through a transition metal complex.
Table 2: Bond valence sum calculations of 1.
|
Atoms
|
Bond Length
|
Bond Valence
|
Bond valence sum
|
|
O5
|
Mo2
|
2.384
|
0.275
|
0.275
|
|
O11
|
Mo4
|
2.418
|
0.251
|
0.485
|
|
|
Mo3
|
2.445
|
0.234
|
|
|
O16
|
Mo5
|
2.388
|
0.273
|
0.273
|
|
O22
|
Mo6
|
2.388
|
0.273
|
0.273
|
|
O42
|
Mo12
|
2.426
|
0.246
|
0.246
|
|
O46
|
Mo13
|
2.328
|
0.321
|
0.321
|
|
O55
|
Mo16
|
2.377
|
0.281
|
0.281
|
|
O58
|
Mo18
|
2.25
|
0.396
|
0.790
|
|
|
Mo17
|
2.252
|
0.394
|
|
Figure 1: Thermo gravimetric analysis of 1.
Figure 2: {Mo36} clusters interconnected by copper hydrate {Cu(H2O)4} complexes to form 1D chains
Figure 3: A ball and stick view of {Mo36}cluster anion
Cu3Mo2O9, 2
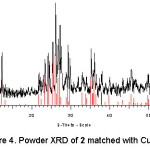
The powder X-ray diffraction pattern (Figure 4) of Cu3Mo2O9, 2, is isostructural with the literature reports.27-28 We did not succeed in growing suitable single crystals of 2 under our reaction condition. In this structure, copper exhibits both octahedral and square pyramidal geometry and molybdenum is tetrahedrally coordinated. The building block of this structure appears to be the occurrence of a trimeric copper cluster29-31 arising from edge sharing of the two copper square pyramids with an octahedron as shown in Figure 5. The trimeric clusters are further linked to {MoO4} tetrahedra to form the 3D network (Figure 6). Around both the copper atoms, there is a strong Jahn-Teller distortion with considerable extension of the apical bonds in comparison to the equatorial bonds.
Figure 4: Powder XRD of 2 matched with Cu3Mo2O9.
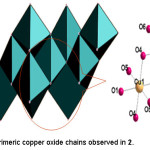
Figure 5: Trimeric copper oxide chains observed in 2.
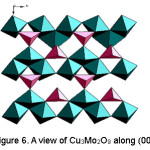
Figure 6: A view of Cu3Mo2O9 along (001).
Magnetic Properties
Organic/inorganic hybrid materials provide new opportunities in the realm of molecular magnetism for studying magnetically condensed (i.e. exchange-coupled) systems. Introduction of secondary metal have shown that the resulting structures are influenced by the coordination preferences of the secondary metal. The magnetism of 1 and 2 may be attributed solely due to the presence of Cu2+ ions (3d9, S=1/2). Since Mo6+ (3d0; S=0) ions do not possess an effective magnetic moment, they do not contribute to the bulk properties. The temperature dependent magnetic susceptibilities of 1 and 2 have been measured in the range 3-300K. All of them follow the Curie-Weiss law at high temperature.
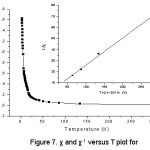
In (NH4)6[{Cu(H2O)4}{Mo36O112(H2O)16}].21H2O, 1, magnetically active centres i.e. Cu(II) are separated by large diamagnetic centres of {Mo36} cluster anions. So the compound is expected to show Curie paramagnetic ordering since there is no magnetic interaction. The thermal variations of χ and 1/ χ of compound are shown in figure 7. The inverse susceptibility plot as a function of temperature is linear, following the Curie- Weiss law with C = 4.1 emu mol-1 and θ =-5.9K. This deviation in Curie and Weiss constants can be due to the presence of paramagnetic impurities in the sample.
Figure 7: χ and χ-1 versus T plot for
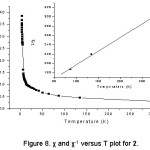
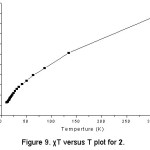
Cu3Mo2O9, 2, consists of copper trimeric chains. The magnetic susceptibility c and 1/c measured in an applied field of 1KOe is shown in figure 8. The thermal variation of cT is shown in figure 9. From 300 to 3 K, cT decreases reaching a minimum at 10 K indicating antiferromagnetic interactions. In the temperature range of 50-300 K, the magnetic susceptibility follows the Curie-Weiss law, c = C / (T – q), with the Curie constant of C = 1.7 emu mol-1 and the Weiss temperature of q = –235K. The negative Weiss temperature suggests the dominance of antiferromagnetic near-neighbor interaction.
Figure 8: χ and χ-1 versus T plot for 2.
Figure 9: χT versus T plot for 2.
Conclusion
It is observed that heteroatom plays two roles: Firstly it links the POM cluster anions and secondly it can act as a charge compensator. The present compounds suggest that a vast family of open framework molybdenum oxide hybrid materials may be assembled by appropriate use of metal ion bridging groups coupled with judicious adjustment of reaction pH, solvents and use of mineralisers. It appears that this rationale of crystal engineering provides a facile method for structural modification of inorganic composite materials and, consequently, tuning of their chemical and physical properties.
Acknowledgement
The authors wish to thank Department Chemistry, IIT Delhi for providing smart apex CCD single crystal X-ray diffractometer under FIST and also MME Department, RGUKT Basar for providing powder X-ray diffractometer. Authors specially thank to Department of Physics and Astronomy, Rowan University, Glassboro, New Jersey, USA for providing magnetic data.
References
- Kera Y., Oonaka T., Yamanaka K., Hirayama S., Kominami H. Highly dispersed transition-metal-containing polyoxomolybdates [XMo6O24n-; X=Fe, Co and Ni] on alumina modified with a silane agent and their catalytic features for partial-methanol-oxidation. Applied Catalysis A. 2004;276:187-195.
CrossRef
- Brito J. L., Barbosa A. L. Effect of Phase Composition of the Oxidic Precursor on the HDS Activity of the Sulfided Molybdates of Fe(II), Co(II), and Ni(II). Journal of Catalysis. 1997;171:467-475.
CrossRef
- Miller J., Sault A. G., Jackson N. B., Evans L., Gonzales M. The formation of active species for oxidative dehydrogenation of propane on magnesium molybdates. Catalysis Letters. 1999;58:147-152.
CrossRef
- Pope M. T., Müller A. Polyoxometalate Chemistry from Topology via Self-Assembly to Applications. Kluwer academic publishers. 2001;23.
- Müller A., Botar B., Das S. K., Bogge H., Schmidtmann M., Merca A. On the complex hedgehog-shaped cluster species containing 368 Mo atoms: simple preparation method, new spectral details and information about the unique formation. Polyhedron. 2004;23:2381-2385.
CrossRef
- Müller A., Kogerler P. From simple building blocks to structures with increasing size and complexity. Coordination Chemical Reviews. 1999;82:3-17.
CrossRef
- Müller A., Roy S. En route from the mystery of molybdenum blue via related manipulatable building blocks to aspects of materials science. Coordination Chemical Reviews. 2003;245:53-166.
CrossRef
- Müller A., Das S. K., Krickemeyer E., Kuhlmann C., Sadakane M., Dickman M. H., Pope M. T. Polyoxomolybdate Clusters: Giant Wheels and Balls. Inorganic Syntheses. 2004;34:191-199.
- Dong-feng L., De-xi Y., Shu-an L., Wen-xia T. The encapsulation of [Cu(en)2]2+ (en=ethylenediamine) by a novel three-dimensional CuII–MoIV bimetallic porous coordination polymer containing zigzag-ladder structure. Inorganic Chemistry Communications. 2002;5:791-795.
CrossRef
- Mukhopadhyay S., Chatterjee P. B., Mandal D., Mostafa G., Caneschi A., Slageren J. V., Weakley T. J. R., Chaudhury M. Honeycomb Nets with Interpenetrating Frameworks Involving Iminodiacetato−Copper(II) Blocks and Bipyridine Spacers: Syntheses Characterization and Magnetic Studies. Inorganic Chemistry. 2004;43:3413-3420.
CrossRef
- Lu C. Z., Wu C. D., Lu S. F., Liu J. C., Wu Q. J., Zhuang H. H., Huang J. S. A three-dimensional zeolite-like organic–inorganic hybrid material constructed from {CuMo2O8N}n double helical chains linked via [Cu(4,4′-bpy)]n fragments. Chemical Communications. 2002;152-153.
CrossRef
- Hagrman P. J., Hagrman D., Zubieta J. Organic-Inorganic Hybrid Materials: From “Simple” Coordination Polymers to Organodiamine-Templated Molybdenum Oxides. Angewandte Chemie International Edition. 1999;38:2639-2684.
CrossRef
- Hagrman D., Zapf P. J., Zubieta J. Organic–inorganic composite oxide phases: one-dimensional molybdenum oxide chains entrained within a three-dimensional coordination complex cationic framework in [{Cu2(triazolate)2(H2O)2}Mo4O13]. Chemical Communications. 1998;1283-1284.
CrossRef
- Hagrman P. J., Zubieta J. Solid-State Coordination Chemistry of Metal Oxides: Hydrothermal Synthesis and Structural Characterization of o-Phenanthroline-Ligated Copper− and Zinc−Molybdenum Oxides. Inorganic Chemistry. 1999;38:4480-4485.
CrossRef
- Hagrman D., Hagrman P. J., Zubieta J. Solid-State Coordination Chemistry: The Self-Assembly of Microporous Organic–Inorganic Hybrid Frameworks Constructed from Tetrapyridylporphyrin and Bimetallic Oxide Chains or Oxide Clusters. Angewandte Chemie International Edition. 1999;38:3165-3168.
CrossRef
- Hagrman D., Haushalter R. C., Zubieta J. Three-Dimensional Organic/Inorganic Hybrid Materials Constructed from One-Dimensional Copper Diamine Coordination Polymers Linked by Bridging Oxoanion Tetrahedra: [Cu(dpe)(MoO4)] and [Cu(dpe)(SO4)(H2O)] (dpe = 1,2-trans-(4-pyridyl)ethene). Chemistry of Materials. 1999;10:361-365.
CrossRef
- Duraisamy T., Ramanan A., Vittal J. J. Two new inorganic-organic hybrid salts: [NH2Me2]4[NMe4]2-[Mo0.5O4⊂(Mo12O36)]·1.5H2O and [NMe4]2[NH4]2[Mo8O26]·2H2O. Journal of Materials Chemistry. 1999;9(3):763-767.
CrossRef
- Upreti S., Ramanan A. The first examples of benzidinium cations templated low-dimensional molybdates. Inorganic Chimica Acta. 2005;358:1241-1246.
CrossRef
- Upreti S., Ramanan A. Structure Directing Role of Hydrogen bonded Dimers: Supramolecular Assemblies of Octamolybdate based Organic/Inorganic Hybrids. Crystal Growth & Design. 2005;5:1837-1843.
CrossRef
- Upreti S., Ramanan A. Role of hydrogen-bonded interactions in the crystal packing of phenylenediammonium phosphomolybdates. Crystal Growth and Design. 2006;6:2066-2071.
CrossRef
- Sheldrick G. M. Acta Crystallography. 1990;46:467.
CrossRef
- Sheldrick G. M. SHELXTL-NT, version 6.12, reference Manual. University of Göttingen, Germany. 2000.
- Wenbin Y., Canzhong L., Xiaoping Z., Quanzheng Z., Jiuhui L., Yaqin Y. A New Member of Giant Polyoxomolybdate Containing Bridging Hydroxylamine Moieties Synthesis and Crystal Structure of (NH4)4Na2[MoVI 36O108(NH2OH)2(OH)6(H2O)12]⋅35H2O. Journal of Cluster Science. 2003;14(3):391-403.
CrossRef
- Guang L., Yong-Ge W., Qing Y., Qun L., Shi-Wei Z. Polyoxometalate chain-like polymer: the synthesis and crystal structure of a 1D compound, [Mo36O108(NO)4(MoO)2La2(H2O)28]n·56nH2O. Inorganic Chemistry Communications. 1999;2(9):434-437.
CrossRef
- Natalia V. I., Maxim N. S., Denis G. S., Alexander R., Dmitrij Y. N., Dieter F., Vladimir P. F. One- Two-and Three-Dimensional Coordination Polymers Built from Large Mo36-Polyoxometalate Anionic Units and Lanthanide Cations. European Journal of Inorganic Chemistry. 2005;(24):4985-4996.
CrossRef
- Chuande W., Xiang L., Wenbin Y., Canzhong L., HongHui Z. Synthesis and Crystal Structure of [(NH4)6][Gd2Mo36O112(H2O)20]·52H2O. Jiegou Huaxue. 2001;20(5):358-362.
- Carmen C., Franca M., Roberto S., Daniele C., Pietro M., Giovanni P. Electron paramagnetic resonance characterisation of silica-dispersed copper molybdate obtained by sol-gel and impregnation methods. Journal of Materials Chemistry. 1999;9(2):07-513.
CrossRef
- Nassau K., Shiever J. W. Cupric Oxide-Molybdenum Oxide Phase Diagram in Air and in Oxygen. Journal of American Ceramic Society. 1969;52(1):36-40.
CrossRef
- Vilminot S., Plouet M. R., Andre G., Swierezynski D., Guillot M., Vigneron F. B., Drillon M. Magnetic structure and properties of Cu3(OH)4SO4 made of triple chains of spins s=1/2. Journal of Solid State Chemistry. 2003;170(2):255-264.
CrossRef
- Radu B., Vincent C., Valerie P., Bernard R. Copper Hydroxydiphosphate with a One-Dimensional Arrangement of Copper Polyhedra: Cu3[P2O6OH]2. Inorganic Chemistry. 2005;44(7):2376-2380.
CrossRef
- Dias H. V. R., Diyabalanage H. V. K., Eldabaja M. G., Elbjeirami O., Omary M. A. R., Omary M. A. Brightly Phosphorescent Trinuclear Copper(I) Complexes of Pyrazolates: Substituent Effects on the Supramolecular Structure and Photophysics. Journal of American Chemical Society. 2005;127(20):489-7501.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.
 , V. Ajay Kumar2
, V. Ajay Kumar2  and N. Laxman2
and N. Laxman2 Material Science Research India An International Peer Reviewed Research Journal
Material Science Research India An International Peer Reviewed Research Journal