Recent Trends in Chitosan Nanofibers: From Tissue-Engineering to Environmental Importance: A Review
Saima Wani1 , Hasham S. Sofi2
, Hasham S. Sofi2  , Shafquat Majeed2
, Shafquat Majeed2  and Faheem A. Sheikh2*
and Faheem A. Sheikh2* 
1Department of Biochemistry, Sher-e-Kashmir University of Agricultural Sciences and Technology, Shalimar Campus, Srinagar-190025, Jammu and Kashmir, India
2Department of Nanotechnology, University of Kashmir, Hazratbal, Srinagar-190006, Jammu and Kashmir, India
Corresponding author Email: faheemnt@uok.edu.in
DOI : http://dx.doi.org/10.13005/msri/140202
Article Publishing History
Article Received on : 10 Nov 2017
Article Accepted on : 29 Nov 2017
Article Published : 25 Nov 2017
Plagiarism Check: Yes
Article Metrics
ABSTRACT:
Chitosan is a biodegradable, biocompatible and extracellular matrix mimicking polymer. These tunable biological properties make chitosan highly useful in a wide range of applications like tissue-engineering, wound dressing material, controlled drug delivery system, biosensors and membrane separators, and as antibacterial coatings etc. Moreover, its similarity with glycosaminoglycans makes its suitable candidate for tissue-engineering. Electrospinning is a novel technique to manufacture nanofibers of chitosan and these nanofibers possess high porosity and surface area, making them excellent candidates for biomedical applications. However, lack of mechanical strength and water insolubility make it difficult to fabricate chitosan nanofibers scaffolds. This often requires blending with other polymers and use of harsh solvents. Also, the functionalization of chitosan with different chemical moieties provides a solution to these limitations. This article reviews the recent trends and sphere of application of chitosan nanofibers produced by electrospinning process. Further, we present the latest developments in the functionalization of this polymer to produce materials of biological and environmental importance.
KEYWORDS:
Chitosan; Nanofiber; Electrospinning; Tissue-engineering; Biomedical
Copy the following to cite this article:
Wani S, Sofi H. S, Majeed S, Sheikh F. A. Recent Trends in Chitosan Nanofibers: From Tissue-Engineering to Environmental Importance: A Review. Mat.Sci.Res.India;14(2)
|
Copy the following to cite this URL:
Wani S, Sofi H. S, Majeed S, Sheikh F. A. Recent Trends in Chitosan Nanofibers: From Tissue-Engineering to Environmental Importance: A Review. Mat.Sci.Res.India;14(2). Available from: http://www.materialsciencejournal.org/?p=6358
|
Introduction
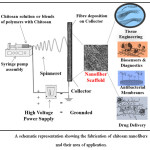
The age old fact that, “ultrathin nanofibers could be drawn from viscoelastic fluids under the influence of external electric field” culminated finally into an ingenuous, handy and simple technique called electrospinning. The developments in this field have started gaining importance since 1993, when Darrell H. Reneker presented his paper highlighting the unexampled features of electrospinning as a fabrication technique, resulting in well and distinct morphology of nanofibers.1 From then onwards, enormous data has been published in this field of engineering science. The electrospinning apparatus is a simple apparatus and consists of four major components: a high voltage power supply, a spinneret (usually a hypodermic needle), a syringe and a collector.2,3 The technique of electrospinning is based on the principle that when a sufficiently high voltage is applied to a spherical droplet of a viscoelastic fluid pumped out through a spinneret, the surface becomes charged, and the repulsion between these charges counteract the surface tension of the spherical droplet leading to destabilization, which further results in nanofiber formation. Moreover, when the repulsion is strong enough between the polymer chains in the droplet, it overcomes the surface tension and is deformed into a conical shape called as Taylor cone.1,4 Afterwards, a jet emancipates from the Taylor cone and because of a combined effect arising from electric field and repulsion among the surface charges, the jet continues to decrease in diameter and finally bends5. In this stage, the jet is said to enter in “whipping instability” regime.6,7 In this regime, the jet accelerates in a whipping motion and the diameter decreases drastically over a period of time, while the solvent keeps on evaporating.7 Finally, the ultrathin jet solidifies and is deposited on the collector screen which is plugged into the opposite charge of the applied field. During the flight, the jet dries and the current is said to change from ohmic flow to convective flow, and the charge migrates to the surface of the fiber.8 Electrospun mats have fiber-like appearance with a diameter range of 100 to 1000 nm or can be more or less than that.9,10
The de-acetylated derivative of chitin also called chitosan is a polysaccharide abundantly found in nature in the shells of crustaceans and functions like collagen in higher vertebrates. This polymer plays an important role in the delivery of drugs and is highly useful in tissue-engineering applications. Chitosan has been found to exhibit synergistic effect along with the anticancer agents and antimicrobials11. Its similarity with glycosaminoglycans makes its suitable candidate for tissue-engineering with excellent biocompatibility, low immunogenicity (though being a TLR agonist), biodegradability, inherent microbial properties, and good mechanical strength. The solubility of chitosan is however a challenge for its widespread applications, and often its fabrication requires harsh solvents.12 These problems of chitosan like solubility, immunogenicity, and stability are improved by thermal, covalent, and non-covalent cross-linking.13–16 This review presents the recent advances in the electrospun nanofibers of chitosan either pristine, or along with other synthetic polymers, and its application in various biomedical fields.
Figure 1
Recent advances in the development of chitosan nanofibers
Recently conducted studies on chitosan and chitosan blended with other polymers have found their utility in various biomedical and analytical fields like tissue-engineering, bone regeneration, nanocatalysis, analytics, filter aids, drug delivery, antibacterial agents, 3D cell culturing and in wound healing materials.
Methodology for the fabrication of chitosan nanofibers
For fabricating the pristine chitosan nanofibers, in most of the work carried out so far, the acetic acid has been used as a single solvent in the electrospinning process. The desirable voltage is 10-15 KV, the tip to collector distance is set at 10-20 cm, and the flow rate is kept 0.1-0.4 ml/hr. Moreover, for the electrospinning of chitosan nanofibers integrated with other polymers (i.e., PLA, PCL, PEO and Silk fibroin etc.), the combination of other solvents like hexafluoro-2-propanol, trifluoroacetic acid, dimethyl carbonate, formic acid and deionized water have been used to solubilize the polymers for electrospinning. Furthermore, after electrospinning, the nanofibers were easily peeled out from the aluminium foil used to cover the collecting drum and/or metallic flat-bed collector. Lastly, the nanofiber mats were dried for minimum 24 h under vacuum in the presence of P2O5 or through the simple drying process in order to get rid of any possible traces of residual moisture. Finally, the samples were characterized and studied for their various applications.
Chitosan nanofibers in tissue-engineering
Arterial tissue-engineered vascular grafts of chitosan and polycaprolactone (PCL) fabricated by electrospinning process had the potential to replace small diameter arterial prosthetic grafts by providing a biodegradable scaffold, where the patient’s own cells can be grafted to form functional neo-tissue. In these studies, the graft was advantageous compared to slow degrading materials used for stents as it provided quick host cell infiltration and remodelling. Moreover, about 1 mm and 5 mm diameter tissue-engineered vascular grafts of chitosan and PCL were implanted in mice as an unseeded infrarenal abdominal aorta interposition conduit for a period of 6 months. After sacrificing these animals, histological analysis demonstrated deposition of extracellular matrix constituents including elastin and collagen, endothelialisation, and organized contractile smooth muscle. It was observed that the mechanical properties of these biodegradable implants were comparable to the native carotid artery. Significant positive correlation between the wall thickness of these grafts and CD68+ macrophage infiltration was attributed to the fast degradation rate of chitosan. Modulation of host macrophage infiltration was the key in reducing excessive neo-tissue formation and stenosis. Successful studies of this arterial graft were conducted in sheep model also17. Hybrid nanofibers composed of chitosan, gelatin, PCL and glutaraldehyde cross-linked were fabricated and tested for in-vitro cell adhesion, growth, and morphology of human foetal fibroblasts (cell line HFFF2). The scaffolds showed hydrophilic character and, satisfactory growth and proliferation, of cell population. Extrapolation of these studies to animals can prove beneficial for these scaffolds to be used as templates for skin tissue-regeneration18. In the field of respiratory tissue-engineering, various scaffolds have been synthesized that mimic the actual respiratory airway to restore the damaged respiratory tree. Depolymerised chitosan and PCL have been electrospun into an elastic and ductile fiber scaffolds. These were then seeded with porcine tracheobronchial epithelial (PTBE) cells using air-liquid interface culture technique that mimics the natural conditions of human airway. The fibrous scaffold of these polymers was found to demonstrate a positive topographical response to PTBE cells, and can be utilized in future as the scaffold material for tracheobronchial tissue-regeneration.19 The presence of another biopolymer with a composite nanobiopolymer fiber results in good mechanical and biological performances including biomineralization. In one of the studies, chitin whiskers enhanced the mechanical properties and osteoblast cell growth of chitosan polyvinyl alcohol (PVA) nanofibers produced by electrospinning. The nanofibers obtained by electrospinning process allowed mineralization of hydroxyapatite in the concentrated simulated fluid. This resulted in the improvement of tensile strength as evidenced by Young’s modulus values, whereas, on the other hand, chitosan whiskers promoted osteoblast cell growth and proliferation. Together, this nano-composite offers a great advantage in bone tissue-engineering applications.20 Bone allografts suffer from the problem of impaired and substandard healing due to loss of periosteum. Artificial membranes fabricated from polysaccharides (chitosan and heparin), and grafted over the bone allografts resemble the natural periosteum. These engineered nanofibers membranes were studied in mouse femur defect model and delivered fibroblast growth factor (FGF-2), transforming growth factor-β1 (TGF-β1), and adipose-derived mesenchymal stem cells (ASCs) to these fractured bones. The results were quite promising, as implanted ACSs responded very well to the engineered periosteum, resulting in the proliferation of these stem cells, in the defective area of femur region. Increased bone callus formation was observed in regions implanted with ASCs as compared to their cell-free controls, while as periosteal cartilage and bone formation was observed in allografts delivering FGF-2 and TGF-β1.21 Incorporation of natural products and/or extracts from herbal materials with nanofibers has led to better mechanical and biological properties of electrospun nanofiber mats. For example, henna extract incorporated into the nanofibers of chitosan and polyethylene oxide (PEO) mats produced by electrospinning process were highly porous, bead-free and showed enhanced antimicrobial properties. These nanofibers showed substantial antibacterial activity against both gram-positive and gram-negative bacteria during in-vitro analysis.22
Chitosan nanofibers as drug delivery systems
Cross-linked three-dimensional nanofiber mats of chitosan and PVA were electrospun to develop a wound healing scaffold. The scaffold contained tetracycline hydrochloride as an antibiotic to prevent wound-associated infections for a longer period of time. In this regard, the tetracycline hydrochloride released from this scaffold showed a burst release delivery initially for a period of 2 h maintaining the inhibitory effect on Staphylococcus epidermis and Staphylococcus aureus. Tetracycline was also found to affect fiber morphology, decreasing the fiber diameter. The exposure of glutaraldehyde vapour increased the cross-linking and provoked a decrease in surface porosity while as raising mat roughness. MTT cell viability and scratch tests showed that, this tetracycline loaded nanofiber mats were not cytotoxic and are good candidates for tissue repairing and wound healing.23 Chitosan nanofibers have been tried in-vivo for delivery of donepezil, a centrally acting reversible acetylcholinesterase inhibitor for quick delivery in Alzheimer’s disease. The fibers were prepared by electrospinning process and then examined under in-vitro and in-vivo animal models. The fibers of chitosan demonstrated a rapid release of donepezil up to 97% in 10 minutes compared to nanofilm of chitosan prepared by ionic gelation. In contrast to other formulations, the in-vivo studies of nanofibers showed maximum absorption as evidenced by Cmax and t1/2 values.24 Transdermal drug delivery of ampicillin sodium was achieved by loading the drug in the fibrous scaffold of a composite polymer of chitosan and PVA fabricated by electrospinning process. The matrix cross-linking by glutaraldehyde improved tensile strength, thermal properties, and hydrophobicity due to cross-linked network between the composite polymers. The ampicillin release from the scaffold fits with the Fickian diffusion showing the initial burst release, followed by sustained release of the drug. The kinetic analysis of the release showed that the drug release fits with the Korsmeyer-Peppas model.25 Genistein, a naturally occurring isoflavone possesses anticancer properties, anti-inflammatory, and topoisomerase inhibitory activities. This compound has been incorporated in a composite electrospun fiber of chitosan and PVA (30:70). Post active and passive loading of genistein on these nanofibers demonstrated 21% progressive release after 30 minutes and 69% release after 24 hours, suggesting a delayed release of the drug, which is essential in certain cases of drug delivery. Such a system has the potential to be used in various biomedical applications like delivery of cancer therapeutics.26 Enzymatic process in the gastrointestinal tract limits the bioavailability of insulin delivery via oral route. One of the possible solutions to this problem is to bypass the gut system to prevent insulin from enzymatic degradation. One of the methods is to deliver the insulin via buccal mucosa which is highly vascularized and permeable, however, the epithelial cells of the buccal system contain tight junctions and hence making diffusion a limiting process. Chitosan-based fibrous nanomats were electrospun along with PEO to incorporate Insulin for oral delivery via buccal mucosa. Insulin permeation across buccal mucosa was studied in an ex-vivo porcine transbuccal model. It was found that, insulin release from these nanomats showed 16 times higher permeability as compared to free insulin, which makes these nanofibers excellent delivery systems for oral delivery of insulin.27 Chitosan-phospholipid nanofiber system was developed recently to demonstrate the release of certain drugs like diclofenac, vitamin B12, and curcumin through transdermal drug delivery systems. This scaffold was biocompatible and fibroblast cell line (L929) grown on these fibers showed similar metabolic activity as compared to cells grown on tissue culture plate. During these studies, it was demonstrated that vitamin B12 has a higher release as compared to diclofenac and curcumin.28 In the field of cell imaging and cancer mitigation, functionalized nanofibers have been used which can perform dual functions. Eryun Yan et al. composed electrospun nanofibers of chitosan and PVA containing gold nanorods. Gold nanorods incorporated into these nanofibers offer applications for imaging with optical microscopes due to their strong plasmon-enhanced absorption and high light-scattering ability. This can provide information about intracellular locations by combining electronic and optical microscopies. Incorporation of anti-cancer agents like doxorubicin with these functionalized nanofibers is hypothesized to offer dual functions, and, act as targeted drug delivery systems delivering anticancer drugs to the nucleus.29 Blends of chitosan and PVA were fabricated along with curcumin functionalized into graphene oxide and silicon. The delivery system mat demonstrated antibacterial and anti-cancer properties of curcumin in a sustained release profile with an initial burst phase. The superior potency of the drug was observed from these mats especially against MRSA and Staphylococcus epidermis infections. The in-vitro cell toxicity experiments on MCF-7, HEPG2 and L929 cell line suggested that these functionalized nanofiber mats are promising candidates for postoperative chemotherapy.30
Asiaticosides derived from Centella Asiatica is a triterpenoid possessing wound healing and anti-inflammatory properties. It has been electrospun with electrostatically interacting polymers, chitosan and sodium alginate using core-shell co-axial electrospinning. The composite nanofibers have been tested in-vivo in Sprague-Dawley rats with circular deep partial thickness burn injuries. The asiaticoside loaded composite nanofibers showed faster drug release profile, and this facilitated the wound healing process, as demonstrated by the improvement in wound healing ratio and pathological sections. Expression of markers of improved wound healing processes like vascular endothelial growth factor (VEGF), cluster of differentiation 31 (CD31), and proliferating cell nuclear antigen (PCNA), and down-regulation of tumor necrosis factor (TNF) and interleukin-6 (IL-6) clearly indicated improvement in wound healing process. All the results demonstrate that these fibers can prove to be useful biomaterials for deep partial burn injuries.31
Chitosan nanofibers in wound healing regeneration
Chitosan was electrospun along with arginine to form a highly porous and hydrophilic wound healing dressing that mimics native skin properties. The human fibroblasts adhered and proliferated in contact with these membranes suggesting biocompatibility. These nanofibers mats also suggested bacteriostatic activity against Escherichia coli and Staphylococcus aureus. The in-vivo application of these mats on Wistar rats showed improved tissue-regeneration and faster wound closure, when compared to pristine nanofibers of chitosan. Ketamine anaesthetised rats were burn inducted after shaving and treated with chitosan-arginine and/or only chitosan scaffolds. Wound healing process was followed using digital camera and analysis of pre-treated and post-treated wounds were done by Image J. software. Necropsy was performed to collect the regenerated tissue and the major organs for histological analysis. It was demonstrated that these membranes significantly improved tissue-regeneration of full-thickness wounds.32 Electrospun composite fibers of chitosan and pullulan fabricated along with tannic acid showed a synergistic antimicrobial activity against gram-negative Escherichia coli. To enhance applicability in wound dressing materials, these membranes were cross-linked, and were shown to exhibit good water absorption ability. These membranes allowed growth of fibroblasts by providing extracellular matrix (ECM) mimicking environment in the skin and allowed interlayer growth of cells. These materials have the potential to be used in deep healing wounds.33
Chitosan nanofibers as antibacterial systems
Formation of tiny pockets between gums and teeth, gingival inflammation, and degradation of gums, supporting structures like ligaments are the characteristic features of Periodontitis. The pathological condition is usually treated by mechanical intervention and systemic antimicrobial therapy of high oral doses. A green route for synthesising Ag nanoparticles (NPs) on the surface of chitosan-PEO nanofibers, involves reducing silver nitrate (AgNO3) with Falcaria vulgaris herbal extract. The addition of this dark green plant extract turns the silver nitrate solution dark-grey, indicating the formation of Ag NPs. These NPs were then loaded on electrospun nanofibers mats of chitosan and PEO for wound dressing applications. The bioactive Ag NPs revealed 100% bactericidal activities against both Staphylococcus aureus and Escherichia coli. Initially, for 8 hours the Ag NPs were sharply released from the nanofiber mats and, after that phase, its release was constant.34 Novel studies have demonstrated that electrospinning of natural polymers with essential volatile oils enhances the antimicrobial efficacy. In one such study, cinnamaldehyde which is an essential oil and eradicates pathogens non-specifically was incorporated into electrospun nanofibrous mats of chitosan and PEO. Cinnamaldehyde in a concentration range of 0.5 to 5% was incorporated into these mats with a fiber diameter of ~ 50 nm. It was found during release studies that cinnamaldehyde was released both in the form of vapours and liquid and that the cinnamaldehyde vapour release over a period of 180 min was two fold greater than cinnamaldehyde liquid release from these composite mats. High inactivation rates against Escherichia coli and Pseudomonas aeruginosa were observed, which can be attributed to the synergistic effect of chitosan and cinnamaldehyde activity. This fibrous scaffold can prove as a potential mat for delivering the broad-spectrum natural antimicrobial agent, cinnamaldehyde, for treatment of nosocomial infections.35 Antibacterial dressing material has been fabricated using nanofibrous mats for simultaneous wound healing along with antibacterial properties. Implantable devices have been mostly associated with percutaneous infections, and using antibacterial wound dressing implantable materials can overcome this problem. Chitosan and polyethylene oxide were electrospun along with chlorhexidine and reduced Ag NPs for dual delivery of these antimicrobial agents. Morphological studies of these electrospun nanofibers show homogenous distribution of Ag NPs throughout the fibers. The release studies of these fibers revealed that chlorhexidine was released for 2 days, while Ag NPs were released for a period of 28 days. Long-term antibacterial efficacy against Staphylococcus aureus was observed over a period of 4 days using 5% AgNO3 solution.36 Synthetic derivatives of chitosan like thiol-chitosan and chitosan iodoacetamide, synthesised by treating chitosan with thioglycollic acid and iodoacetic acid, respectively, and cross-linking by glutaraldehyde showed morphologically greater stability than chitosan or chitosan blended nanofibers. These derivatives also displayed superior antibacterial activity against gram-negative bacteria Escherichia coli with the minimum inhibitory concentration of 400 mg/ml. The thiol groups being potent nucleophiles increase the antibacterial activity through disulphide bond formation with the proteins in the outer cell membrane of the bacterial cell.37
Chitosan nanofibers as biosensors, analytic systems and diagnostic aids
Nanofibers of natural polymers find their applications even in the field of chromatographic analysis. Chitosan-Fe nanofiber composites were synthesized by electrospinning and applied for mat-based extraction of trace amount of 9-tetrahydrocannabinol from whole blood sample following its detection by UV detectors. The high surface area of these composite natural nanofibers ensured strong interaction between the adsorbent and the analyte. Compared to the adsorbent in C18 columns, this new sorbent could offer better analytical performance in terms of recovery, reproducibility and detection limit with low consumption of hazardous organic solvents and sorbent material.38 Nanofibers because of spatial structure, high porosity and large surface area allow enzymes to be immobilized in this network and can be developed into sensitive electrochemical biosensors. Chitosan and PVA electrospun nanofibers were successfully fabricated into an ultra-sensitive electrochemical biosensor by immobilizing genetically-engineered acetylcholinesterase into these fibers. This enzyme immobilized system was successfully developed for rapid detection of Pirimiphos-methyl in olive oil. The biosensing performance for detection of pirimiphos-methyl improved to a lower limit of detection value (0.2 nM) compared to already existing international set value of 164 nM.39 Fluorescence sensing systems comprising of rhodamine-B decorated on electrospun chitosan nanofibers allow detection of Hg2+ ions present in various analytical samples. These rhodamine decorated chitosan nanofibers allowed selective detection of Hg2+ ions by opening the spirolactam ring of the rhodamine unit. The huge specific surface area provided by nanofibers surface makes these mats as enhanced sensitive sensing probes. This strategic fabrication has the potential to design and develop a high performance sensing material for the detection of various metal elements in the environmental field.40 Electrospun nanofibers find their application in the field of catalysis as well, and have been utilized by a research group recently to catalyse some of the major challenging reactions like reduction of nitro aromatics. In one such study, nickel NPs have been surface decorated on electrospun nanofibers of chitosan and PCL to catalyse the reduction of various nitrophenols like 2-nitrophenol, 2,4-dinitrophenol and 2,4,6 trinitrophenol under mild conditions. This nanocatalyst was synthesized by deposition-reduction method and the catalytic activity was evaluated by the catalytic reduction of nitrophenols in aqueous solutions in the presence of sodium borohydride as reducing agent and Ni decorated chitosan-PVA nanofibers as catalyst. Moreover, this catalytic scaffold has shown a flexible characteristic along with reusability property.41 In the field of tumor diagnosis, nanofiber materials are providing a new platform to isolate and characterize the circulating tumor cells in the blood, considered as biomarkers of cancer metastasis. Electrospun porous nanofibers of chitosan functionalised by poly(carboxybetaine methacrylate) (pCBMA) brushes provide interfacial properties for the surface capture of circulating tumor cells. pCBMA brushes integrated into chitosan nanofiber interface control non-specific cell adhesion and multivalent immobilization of biomolecules like antibodies and aptamers resulting in efficient capture of circulating tumor cells. Also, the DNA aptamers, epithelial cell adhesion molecule (EpCAM), were integrated into the pCBMA brushes to induce highly efficient and specific cell capture. The release of the circulating tumor cells was achieved by binding complementary sequence to hybridize the aptamer.42
Fiber-based biosensor of chitosan and carbon nanotubes covering an electro-deposited layer of Ag NPs on a gold electrode was developed for the determination of uric acid by immobilization of urease enzyme. This amperometric uric acid biosensor sensed uric acid at an optimum potential of -0.35 V based on the reduction of dissolved oxygen, during oxidation of uric acid by the immobilized urease enzyme. This biosensor has a high affinity for uric acid as indicated by the low value of Michaelis-Menton constant of 0.21 mML-1. Further, the presence of other analytes present in the serum samples like glucose, ascorbic acid or lactic acid didn’t interfere in the determination of the uric acid. The scaffold has the potential to be used as a supporting material for biosensor applications involving oxidase enzymes.43
Chitosan as filter media for treatment of contaminated water
Spiral wound chitosan nano-membranes produced by electrospinning of chitosan was studied for treating Cr(VI) contaminated water. The loading capacity of the module was dependent on the flow rate and nanofiber deposition density but independent on Cr(VI) concentration. The slower flow rate of untreated water was related to higher adsorption of Cr(VI) ions. The maximum adsorption capacity obtained in this module with 2 g/m2 nanofiber membranes was 20.5 mg/g at 10% breakthrough. Further, the module could also adsorb Cu(II), Cd(II) and Pb(II) ions separately, but shows good selectivity to Cr(VI), when these metal ions were coexisting together. By comparing the filtration efficiency of these fibers with that of commercial membranes available for adsorption of Cr(VI), it was found that these fibers perform much better in terms of metal ion rejection, permeation flux and the trans-membrane pressure for easy operation while leaving no concentrated water.44 Reusable membranous scaffolds of chitosan and PVA were fabricated by electrospinning process and adsorption capacity of these fibers was tested for Cr(VI), Fe(III), and Ni(II) ions over a wide range of concentrations and time points. The study demonstrated higher adsorption rate of these ions, however less adsorption capacity at high concentrations. The membranes showed reusability even after five runs and could be a potential membrane scaffold for filtration purposes of the metal ions from the low concentrated solutions.45 Lincosamides like Clindamycin is a broad spectrum antibiotic which is hardly metabolized and any metabolite formed still retains activity. Clindamycin causes side effects like stomach pain, rashes, nausea, and vomiting. Natural and industrial water has been found to be contaminated with clindamycin and different techniques like ion exchange, coagulation and adsorption can be utilized to remove it from these sources. Chitosan-Ag2S nanocomposite fibers have been fabricated for the removal of clindamycin by adsorption on these composite fibers. Significantly high adsorption quantities of clindamycin have been reported on these fibers and, make these membranes suitable for removal of clindamycin for drinking water.6
Chitosan nanofibers as supporting material in 3D cell culturing
In bone tissue-engineering, scaffolds of polymers provide the architecture for bone forming cells to adhere, penetrate and subsequently proliferate to form new bone tissue. Electrospinning is a versatile technique to develop such scaffolds that can temporarily act as architects for bone formation during bone injury cases. In one such study, researchers fabricated electrospun nanofibers of chitosan and silk fibroin (biopolymers), and studied the cell growth and proliferation of human mesenchymal stem cells (hMSCs) towards osteogenic differentiation. The in-vitro studies like cell culture analysis, cytoskeleton analysis, MTT assay suggested proliferation of these cells on chitosan-silk fibroin nanofibers. It was also found in these studies that, chitosan guided the differentiation of hMSCs towards osteogenic lineage and induced bone formation.47 The addition of crystal materials has been found to increase the production of fiber and produce uniform fibers. The addition of cellulose nanocrystal to the chitosan-polyethylene oxide solutions allows production of bulk fibers, which are uniform and thermally stable. The high proportion of chitosan fibers results in improvement of biological properties. These nanofibers due to the presence of crystalline cellulose in cultures of 3T3 fibroblasts promote changes in cytoskeleton organisation as evidenced in F-actin and β-tubulin. A 3-D tissue model of the liver was developed using the process of electrospinning. Electrospun nanofibrous scaffolds along with co-culturing hepatocytes and fibroblasts developed into a liver-like tissue showed long-term liver like functions. Fibronectin was adsorbed on these nanofiber scaffolds to enhance adhesion of hepatocytes and fibroblasts. Compared to 2D cultures, it was found that these 3D culture systems formed well-defined colonies and maintained their morphologies for a prolonged period of time, demonstrating high levels of cytochrome activity. 48Laser-ablated ligand-free Au and Si NPs were anchored to the surface of chitosan-polyethylene oxide nanofibers produced by electrospinning. The grafting with bare NPs led to the formation of uniform and cylindrical nanofibers, and offered additional interaction with biological components due to the electrostatic interactions. Spectroscopic analysis of these fibers revealed better thermal stability and advanced therapeutic modalities. Cytotoxicity assays using human keratinocytes cells (HaCaT) revealed that these nanofibers were safe for cell culture and growth.49 Differentiation of human endometrial stem cells (hEnSCs) in the presence of angiogenic factors was studied over the scaffolds of electrospun chitosan/gelatine bioactive glass powders. The results demonstrated that bioactive glass increased the diameter of nanofibers, and 1.5% of its content provided a suitable three-dimensional structure for endothelial cells differentiation. The expression of endothelial markers (CD31, vascular endothelial cadherin) studied during in-vitro studies revealed that these fiber scaffolds could be promising to regenerate endothelial cells needed in blood vessel repair50
Chitosan nanofibers in nerve tissue-regeneration
Enhancement of peripheral and central nerve regeneration can be achieved by using Schwann cells, which provide a supportive environment for the neurite growth through expression of surface ligands, the release of neurotrophic factors as well as the synthesis of ECM. The Schwann cells express various adhesion molecules on the surface and are found to direct the neurite outgrowth. Oriented chitosan nanofibers were fabricated using the process of electrospinning and were studied for Schwann cell alignment and peripheral nerve regeneration. The Schwann cells were found to align along the nanofibers into oriented fibrous sheets exhibiting a Schwann cell column. These Schwann cell columns were tested in-vivo in rat sciatic nerve defect, and it was found that, electrophysiological and functional recovery occurred in time over these oriented fibers. Histological analysis of the treated sciatic nerve revealed sprouting of the myelinated axons followed by axonal maturation. These studies have revealed that, oriented chitosan mesh tubes may prove to be substitutes for autogenous nerve graft51. A research group developed a novel chitosan-intercalated montmorillonite/polyvinyl alcohol (OMMT/PVA) nanofibrous mesh as a microenvironment for the guiding differentiation of human dental pulp stem cells (hDPSCs) towards neuron-like cells. The mesh was prepared through ion exchange reaction between the montmorillonite (MMT) and chitosan. Later on, the PVA solutions containing various concentrations of OMMT were electrospun to form 3D-nanofibrous meshes of OMMT-PVA. The hDPSCs seeded on the prepared scaffolds induced neuronal specific differentiation as determined by the expression of Oct-4, Nestin, NF-M, NF-H, MAP2, and βIII-tubulin; all markers of neural differentiation. Compared to control group, the population of these cells growing on OMMT-PVA nanofibers differentiated significantly into neuron-like cells in all the experimental groups. The study demonstrated the feasibility of these artificial nerve grafts cultured with hDPSCs for regeneration of damaged neural tissue52.
Other applications of chitosan
Furthermore, a new approach developed for the fabrication of mechanically tough aerogel nanofiber system was put forward. In this study, chitosan was subjected to trimethylsilylation to block hydrophilic -NH2 and -OH groups by cross-linking reaction. Supercritical fluid drying systems utilizing CO2/acetone instead of CO2/alcohol was the key factor for the successful silylation to prevent the deprotection of the silyl ether under high pressure. This aerogel material while keeping its nanoscale structural homogeneity intact has the potential applications to be used as a thermal and acoustic insulator53.
Conclusion and Outlook
This review gives a detailed account of fabrication techniques to synthesise novel materials via functionalization of chitosan and/or by blending it with other polymers. The diverse applications of these novel materials include drug delivery of insulin, as anticancer and antibacterial agents, arterial stunts and in wound healing dressings etc. The novelty of these materials comes from the fact that, their utility has been studied in different domains and, they have shown promise in mimicking bio-interface and possess advanced mechanical properties. However, a lot of work is still needed to overcome several limitations of these nanofibers to be used for cell infiltration. Since most of the work reported in the literature is focused on 2D nanofiber mats only, the 3D-nanofiber mats with absolute porosity and full-thickness must be developed for translating them into real tissue-engineering applications. Moreover, this review focusses on the applications of chitosan derived scaffolds in the field of analysis, cancer diagnosis and biosensors. However, the commercialization of these materials is a distinct dream. The rationale for this may be the complex strategy used and low yielding product after the event of electrospinning. Furthermore, same problems exist with the nanofibers fabricated with the intention to be used in environmental applications (e.g., water purification from contaminated sources), 3D cell culturing, nerve-regeneration and in wound healing materials. In summary, we expect that this review will provide the current insights for using chitosan as a biomimetic material for researchers working in the field of nanomaterial science.
Acknowledgements
This work was funded by DST Nano Mission sponsored project (SR/NM/NM-1038/2016) and Science and Engineering Research Board (SERB) research grants (ECR/2016/001429 & ECR/2017/000205).
References
- Doshi J and Reneker D. H. J. Electrostat. 199;35:151–160.
CrossRef
- Sheikh F. A., Ju H. W., Lee J. M., Moon B. M., Park H. J., Lee O. J.,Kim J. H.,Kim D. K., Park C. H., Nanomedicine Nanotechnology. Biol. Med. 2015;11:681–691.
CrossRef
- Sheikh F. A., Ju H. W., Moon B. M., Lee O. J., Kim J., Park H. J., Kim D. W., Kim D., Jang J. E., Khang G. J. Tissue Eng. Regen. Med. 2016;10:209–221.
CrossRef
- Yarin A. L., Koombhongse S., Reneker D. H. J. Appl. Phys. 2001;90:4836–4846.
CrossRef
- Zussman E., Theron A., Yarin A. L. Appl. Phys. Lett. 2003;82:973–975.
CrossRef
- Taylor G. in Proc. R. Soc. London A Math. Phys. Eng. Sci., The Royal Society. 1969;453–475.
- Shin Y. M., Hohman M. M., Brenner M. P., Rutledge G. C. Appl. Phys. Lett. 2001;78:1149–1151.
CrossRef
- Rebouillat S., Lyons M. E. G. Int. J. Electrochem. Sci. 2011;6:5731–5740.
- Thompson C. J.,Chase G. G., Yarin A. L., Reneker D. H. Polymer (Guildf). 2007;48:6913–6922.
CrossRef
- Frenot A.,Chronakis I. S. Curr. Opin. Colloid Interface Sci. 2003;8:64–75.
CrossRef
- Smith J. K., Moshref A. R., Jennings J. A., Courtney H. S., Haggard W. O. Clin. Orthop. Relat. Res. 2013;471:3158–3164.
CrossRef
- Ignatova M., Manolova N., Rashkov I. Macromol. Biosci. 2013;13:860–872.
CrossRef
- Austero M. S., Donius A. E., Wegst U. G. K., Schauer C. L. J. R. Soc. Interface. 2012;9:2551–2562.
CrossRef
- Donius A. E., Kiechel M. A., Schauer C. L., Wegst U. G. K., J. R. Soc. Interface. 2013;10:20120946.
CrossRef
- Schiffman J. D., Schauer C. L. Biomacromolecules. 2007;8:594–601.
CrossRef
- Kiechel M. A., Schauer C. L. Carbohydr. Polym. 2013;95:123–133.
CrossRef
- Fukunishi T., Best C. A.,Sugiura T., Shoji T., Yi T., Udelsman B., Ohst D., Ong C. S., Zhang H.,Shinoka T. PLoS One. 2016;11:e0158555.
CrossRef
- Gomes S., Rodrigues G., Martins G., Henriques C., Silva J. C. Int. J. Biol. Macromol. 2017;102:1174–1185.
CrossRef
- Mahoney C., Conklin D., Waterman J., Sankar J., Bhattarai N. Biomater. Sci. Polym. Ed. 2016;27611–625.
CrossRef
- Pangon A., Saesoo S., Saengkrit N., Ruktanonchai U.,Intasanta V. Carbohydr. Polym. 2016;144:419–427.
CrossRef
- Romero R., Travers J. K., Asbury E., Pennybaker A., Chubb L., Rose R., Ehrhart N. P., Kipper M. J. J. Biomed. Mater. Res. Part A. 2017;105:900–911.
CrossRef
- Yousefi I., Pakravan M., Rahimi H., Bahador A., Z. Farshadzadeh A.,Haririan I. Mater. Sci. Eng. C. 2017;75: 433–444.
CrossRef
- Alavarse A. C., de Oliveira F. W. S., Colque J. T., da Silva V. M., Prieto T., Venancio E. C., Bonvent J. J. Mater. Sci. Eng. C. 2017;77:271–281.
CrossRef
- AnjiReddy K., Karpagam S. Int. J. Biol. Macromol. 2017;105:131–142.
CrossRef
- Cui Z.,Zheng Z., Lin L., Si J., Wang Q.,Peng X., Chen W. Adv. Polym. Technol. 2017.
- Ibrahim S., Sayed H. M., El-Rafei A. M., Amir A. E.,Ismail M., Allam N. K. J. Mol. Liq. 2016;223:1056–1061.
CrossRef
- Lancina M. G., Shankar R. K., Yang H. J. Biomed. Mater. Res. Part A. 2017;105:1252–1259.
CrossRef
- Mendes A. C., Gorzelanny C., Halter N., Schneider S. W., Chronakis I. S. Int. J. Pharm. 2016;510:48–56.
CrossRef
- Yan E., Cao M., Wang Y., Hao X., Pei S., Gao J., Wang Y., Zhang Z., Zhang D. Mater. Sci. Eng. C. 2016;58:1090–1097.
CrossRef
- Sedghi R., Shaabani A., Mohammadi Z., Samadi F. Y., Isaei E. Carbohydr. Polym. 2017;159:1–10.
CrossRef
- Zhu L., Liu X., Du L., Jin Y. Biomed. Pharmacother. 2016;83:33–40.
CrossRef
- Antunes B. P., Moreira A. F.,Gaspar V. M., Correia I. J. Carbohydr. Polym. 2015;130:104–112.
CrossRef
- F. Xu, B. Weng, R. Gilkerson, L.A. Materon, K. Lozano, Carbohydr. Polym. 115 (2015) 16–24.
CrossRef
- Kohsari I., Shariatinia Z., Pourmortazavi S. M. Carbohydr. Polym. 2016;140:287–298.
CrossRef
- Rieger K. A., Schiffman J. D. Carbohydr. Polym. 2014;113:561–568.
CrossRef
- Song J., Remmers S. J. A., Shao J., Kolwijck E., Walboomers X. F., Jansen J. A.,Leeuwenburgh S.C.G.,Yang F. Nanomedicine Nanotechnology. Biol. Med. 2016;12:1357–1364.
- Abdelgawad A. M., El-Naggar M. E., Hudson S. M., Rojas O. J. Int. J. Biol. Macromol. 2017;94:96–105.
CrossRef
- Asiabi M., Mehdinia A., Jabbari A., Chromatogr J. A. 2017;1479:71–80.
CrossRef
- El-Moghazy A. Y., Soliman E. A.,Ibrahim H. Z., Marty J. L.,Istamboulie G., Noguer T. Talanta. 2016;155:258–264.
CrossRef
- N. Horzum, D. Mete, E. Karakuş, M. Üçüncü, M. Emrullahoğlu, M.M. Demir, ChemistrySelect 1 (2016) 896–900.
CrossRef
- K. Karakas, A. Celebioglu, M. Celebi, T. Uyar, M. Zahmakiran, Appl. Catal. B Environ. 203 (2017) 549–562.
CrossRef
- N. Sun, M. Liu, J. Wang, Z. Wang, X. Li, B. Jiang, R. Pei, Small 12 (2016) 5090–5097.
CrossRef
- A. Numnuam, P. Thavarungkul, P. Kanatharana, Anal. Bioanal. Chem. 406 (2014) 3763–3772.
CrossRef
- L. Li, J. Zhang, Y. Li, C. Yang, J. Memb. Sci. 544 (2017) 333–341.
CrossRef
- U. Habiba, A.M. Afifi, A. Salleh, B.C. Ang, J. Hazard. Mater. 322 (2017) 182–194.
CrossRef
- V.K. Gupta, A. Fakhri, S. Agarwal, M. Azad, Int. J. Biol. Macromol. 103 (2017) 1–7.
CrossRef
- G.-J. Lai, K.T. Shalumon, S.-H. Chen, J.-P. Chen, Carbohydr. Polym. 111 (2014) 288–297.
CrossRef
- D. Rajendran, A. Hussain, D. Yip, A. Parekh, A. Shrirao, C.H. Cho, J. Biomed. Mater. Res. Part A (2017).
- A. Al-Kattan, V.P. Nirwan, E. Munnier, I. Chourpa, A. Fahmi, A. V Kabashin, RSC Adv. 7 (2017) 31759–31766.
CrossRef
- A. Shamosi, D. Mehrabani, M. Azami, S. Ebrahimi-Barough, V. Siavashi, H. Ghanbari, E. Sharifi, R. Roozafzoon, J. Ai, Artif. Cells, Nanomedicine, Biotechnol. 45 (2017) 163–173.
CrossRef
- W. Wang, S. Itoh, K. Konno, T. Kikkawa, S. Ichinose, K. Sakai, T. Ohkuma, K. Watabe, J. Biomed. Mater. Res. Part A 91 (2009) 994–1005.
CrossRef
- H. Ghasemi Hamidabadi, Z. Rezvani, M. Nazm Bojnordi, H. Shirinzadeh, A.M. Seifalian, M.T. Joghataei, M. Razaghpour, A. Alibakhshi, A. Yazdanpanah, M. Salimi, ACS Appl. Mater. Interfaces 9 (2017) 11392–11404.
CrossRef
- S. Takeshita, S. Yoda, Nanoscale 9 (2017) 12311–12315.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.
 , Hasham S. Sofi2
, Hasham S. Sofi2  , Shafquat Majeed2
, Shafquat Majeed2  and Faheem A. Sheikh2*
and Faheem A. Sheikh2* 
 Material Science Research India An International Peer Reviewed Research Journal
Material Science Research India An International Peer Reviewed Research Journal